
OpenFOAM-9 multiphaseEulerFoam, wallBoiling, changing table Hs to accommodate new pressure : r/OpenFOAM
Ideal and Real Discharge Coefficients – Using Fundamental Equations of State in Mass-Flow Measurements with Sonic Nozzles R. S
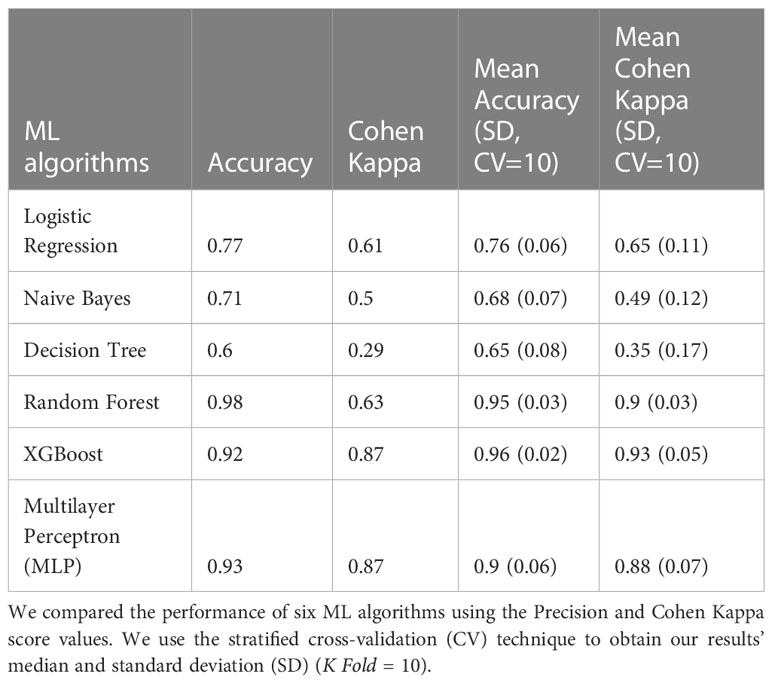
Frontiers | Dysbiosis signatures of gut microbiota and the progression of type 2 diabetes: a machine learning approach in a Mexican cohort

Cp-Cv for real gas, in terms of alpha & beta, change in internal energy with respect to volume - YouTube

Free light chain kappa and the polyspecific immune response in MS and CIS – Application of the hyperbolic reference range for most reliable data interpretation - ScienceDirect

Expression of Nuclear factor-kappa B by immunohistochemical staining.:... | Download Scientific Diagram

The equation is given below, CP=CV+TV(Beta2/K), links CP and CV with Beta and K. Use this equation to evaluate CP - CV for an ideal gas. CP=CV+T(Lambda P Lambda T)V(Lambda V Lambda

SOLVED: The general expression for the difference in the heat capacities is given by: Cp = Cv + T^2KV. Show that for an ideal gas, this expression becomes: Cp = Cv = nR.



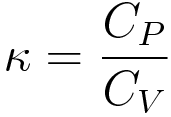


![Question] Composition summation · Issue #347 · usnistgov/REFPROP-wrappers · GitHub Question] Composition summation · Issue #347 · usnistgov/REFPROP-wrappers · GitHub](https://user-images.githubusercontent.com/58614082/103503111-e8fcdf00-4e18-11eb-99f3-f320c36dd40c.png)






