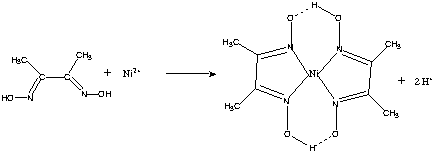
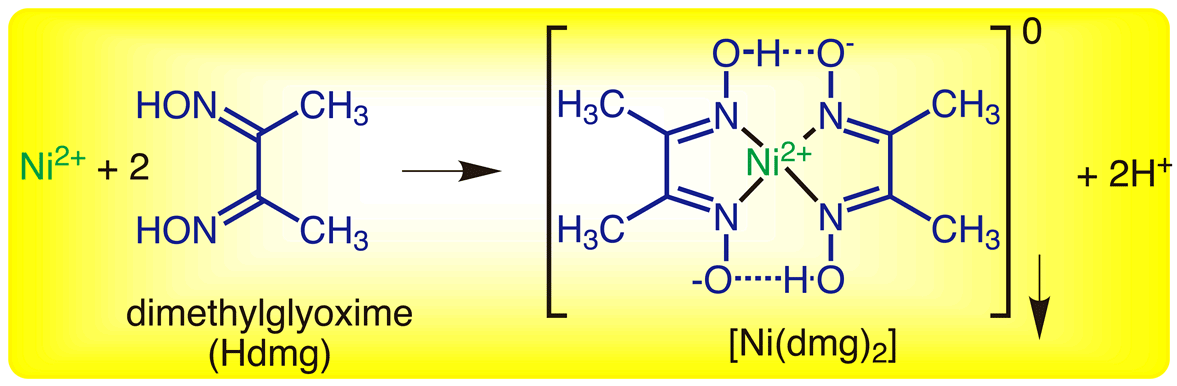
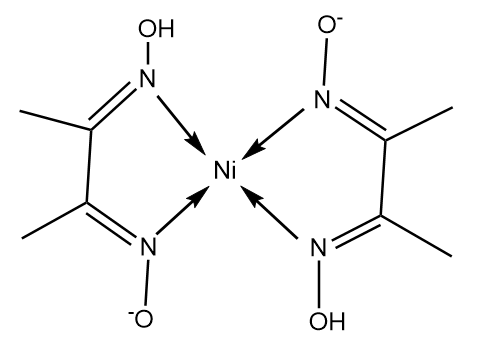
In basic medium the amount of Ni2+ in a solution can be estimated with the dimethylglyoxime reagent. The correct statement(s) about the reaction and the product is(are):
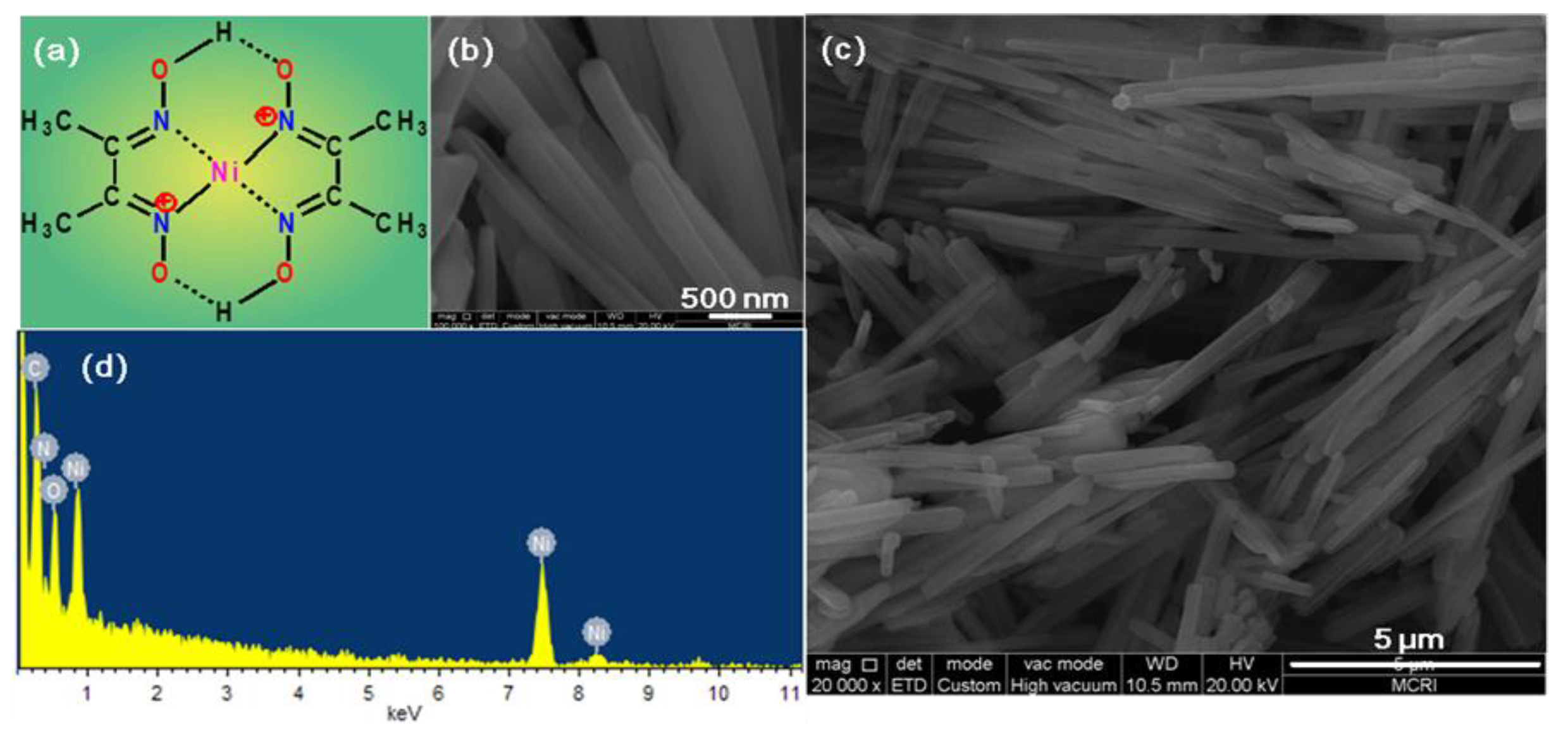
Catalysts | Free Full-Text | Study on Thermal Decomposition Behavior, Gaseous Products, and Kinetic Analysis of Bis-(Dimethylglyoximato) Nickel(II) Complex Using TG-DSC-FTIR-MS Technique

Ultratrace Detection of Nickel(II) Ions in Water Samples Using Dimethylglyoxime-Doped GQDs as the Induced Metal Complex Nanoparticles by a Resonance Light Scattering Sensor | ACS Omega

Given below are two statements:Statement I: The identification of Ni2+ is carried out by dimethylglyoxime in the presence of NH4OHStatement II: The dimethylglyoxime is a bidentate neutral ligand.In the light of the